Why are plants green?

We asked three of our International Undergraduate Summer School students; Poppy Smith, Lyvy Hall and Tom Hammond, why plants are green?
“The short answer is that plants look green to us, because red light is the most useful wavelength for them.
The longer answer lies in the details of photosynthesis, the electromagnetic spectrum, energy and “special pairs” of chlorophyll molecules in each plant cell.
Plants can broadly be divided into four main components: roots, stem, flowers and leaves. The leaves make the plant’s energy, or food in, as every GCSE student knows, the process called photosynthesis.
Plants (plus algae and certain bacteria) absorb light to make sugars, providing the plant with energy and some other useful biochemical products which the plant requires to grow successfully.
Light which can be seen by the human eye (the visible light spectrum) is made up of the rainbow of colours, stretching from purple through to red. Objects are perceived by humans as coloured when the object reflects light back to our eyes. All the other visible wavelengths of light are absorbed, and we only see the reflected wavelengths.
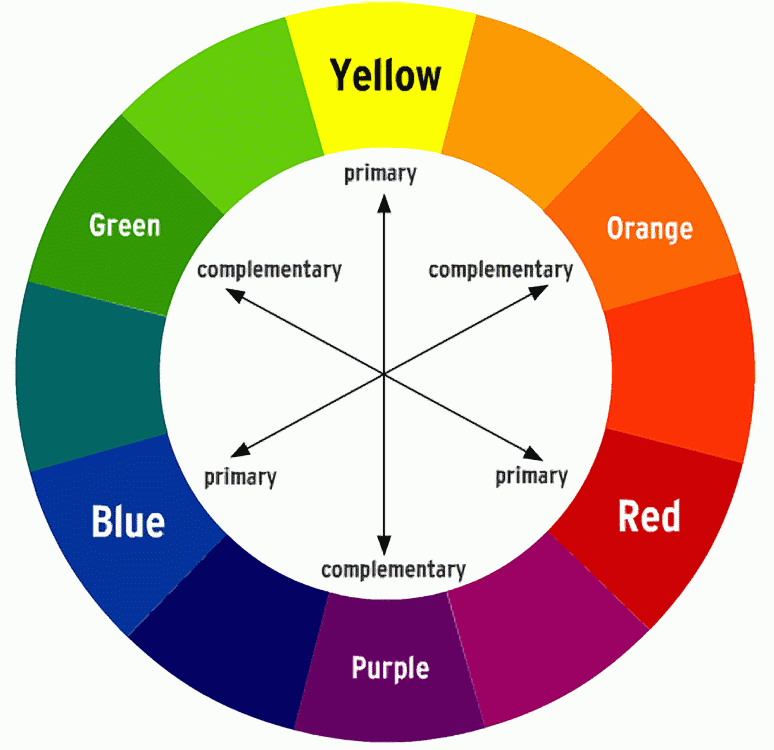
The colours of visible light form a colour wheel. Within that wheel the colour an object appears to be is the colour complementary to the one it most strongly absorbs.
As such, plants look green because they absorb red light most efficiently and the green light is reflected.
Light and the electromagnetic spectrum
Visible light is part of the electromagnetic spectrum, the collection of all light.
Light travels in waves, and thus it has a wavelength, which corresponds to the distance between the peaks of the waves.
Visible light has wavelengths from 380 nanometres for purple, through to 730 nanometres for red. To put this into perspective, a human hair is 100,000 nanometres thick.
Shorter wavelengths have a higher energy, the frequency of the ‘wave’ is higher, so purple light has more energy than red light.
How plants use light
Photosynthesis is essentially the process of the plant converting atmospheric gas carbon dioxide (CO2) and water (H2O) into simple sugars, producing oxygen (O2) as a by-product. To do this, it needs energy and it gets that energy from the light it absorbs.
By absorbing light, the object also absorbs some of the energy carried by the light. In the case of plants, it is the pigment chlorophyll which absorbs the light, and it is picky about which wavelengths it absorbs – mostly opting for red light, and some blue light.
The absorbed energy causes the electrons in the object to become excited.
When electrons are excited, they are promoted from a level of low energy to a level of higher energy. The energy in the light makes the electrons excited and removes energy from the light – this is an example of the first law of thermodynamics – energy is neither created nor destroyed it can only be transferred or changed from one form to another.
That process takes place in specific compartments within cells called chloroplasts and is split into two stages;
- The first stage sees a sequence of reactions which are ‘light-dependent’. Chloroplasts contain many discs called thylakoids, which are packed with chlorophyll. Structures within the thylakoids known as photosystems form the core machinery of photosynthesis and at the centre of each photosystem are a ‘special pair’ of chlorophyll molecules. Electrons in these chlorophyll molecules are excited upon absorption of sunlight. The job of the rest of the chlorophyll molecules in the chloroplast is simply to pass energy towards the special pair
- A second set of reactions are light-independent. These use the energy captured during the light-dependent step to make sugars. These reactions occur in the fluid which bathes the thylakoids (the stroma)
During these reactions, CO2 dissolves in the stroma and is used in the light-independent reactions. This gas is used in a series of reactions which results in the production of sugars. Sugar molecules are then used by the plant as food in a similar way to humans, with excess sugars stored as starch, ready to be used later, much like fat storage in mammals.
Therefore, the red end of the light spectrum excites the electrons in the leaves of the plants, and the light reflected (or unused) is made up of more of wavelengths of the complementary (or opposite) colour, green.
So, plants and their leaves look green because the “special pair” of chlorophyll molecules uses the red end of the visible light spectrum to power reactions inside each cell. The unused green light is reflected from the leaf and we see that light. The chemical reactions of photosynthesis turn carbon dioxide from the air into sugars to feed the plant, and as a by-product the plant produces oxygen.
It is this preference for light at the red end of the spectrum that is behind Dr Brande Wulff and his team’s development of speed breeding technology. The technique first used by NASA to grow crops in space uses extended day-length, enhanced LED lighting and controlled temperatures to promote rapid growth of crops.
It speeds up the breeding cycle of plants: for example, six generations of wheat can be grown per year, compared to two generations using traditional breeding methods.
By shortening breeding cycles, the method allows scientists and plant breeders to fast-track genetic improvements such as yield gain, disease resistance and climate resilience in a range of crops such as wheat, barley, oilseed rape and pea.”

