Iron in plants for human nutrition
Plant foods can be a good source of iron, but it depends on the amount naturally present in different crops and the bioavailability.
For example, seeds and grains store large amounts of iron (and zinc), but they also contain phytic acid – a storage form of phosphorus, which inhibits mineral bioavailability in the gut. In pulses (beans and peas), minerals and phytic acid are evently distributed, but in cereal grains both compounds are concentrated in the outer layers (bran) while being low in the centre of the grain (endosperm).
Our collaborative research has shown that iron and phytic acid can be separated in time or by location. We focus on two major crops, peas and wheat.
- Biofortification of wheat
- Iron in peas
Biofortification of wheat
In a joint effort the laboratories of Janneke Balk, Cristobal Uauy and Wendy Harwood have developed wheat lines with a significant increase in iron concentration in the grain endosperm. After milling, the white flour contains more than 20 mg/kg iron, compared to 5 – 6 mg/kg in normal wheat flour (Connorton et al., 2017; Harrington et al., 2023).
In the UK and many other countries, cereal flours are fortified with iron – usually iron sulphate or iron powder – of at least 16.5 mg/kg*. Thus, the levels we can achieve by minor genetic changes could in theory replace iron fortification and remove it from ingredient labels.
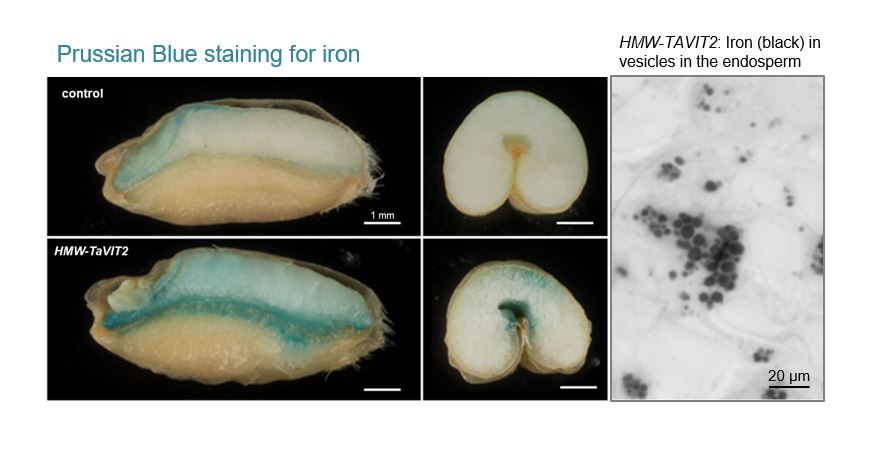
By altering the expression pattern of a gene for a vacuolar iron transporter (VIT), iron is re-distributed from the bran layers to the endosperm. The VIT gene was isolated from wheat, and reinserted in the genome, a process known as a cis-genic rather than transgenic. The wheat lines have successfully undergone field trials in 2019 and 2021 (manuscript in preparation).
In a second set of wheat lines, we have combined the VIT expression cassette with overexpression of a rice nicotianamine synthase gene (NAS). NAS overexpression in wheat was previously trialled by the groups of Navreet Bhullar (Singh et al., 2017) and Alex Johnson (Beasley et al., 2019). The “VIT-NAS” grain produces white flour with 20 – 25 mg/kg iron and slightly elevated zinc, and wholemeal flour with two-fold more zinc, up to 50 mg/kg (Harrington et al., 2023).
As a third line of investigation, we are using gene-editing to target regulatory genes of iron homeostasis. Please come back to this web page in a few months to read how this progresses!
*For the UK Bread and Flour Regulations, see www.legislation.gov.uk/uksi/1998/141/contents/made.
Why make high iron wheat?
How do you make high iron wheat?
Iron in peas
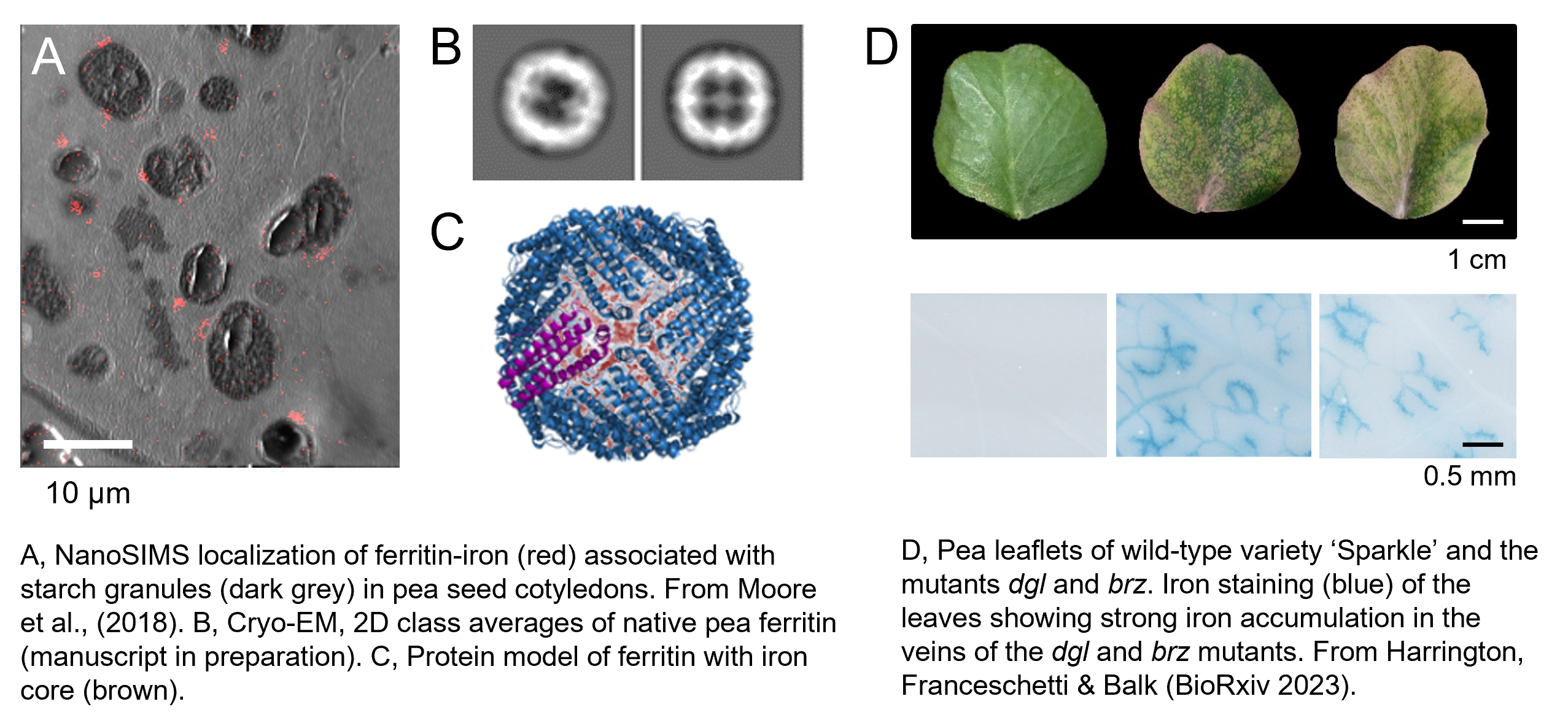
Pulses such as peas and beans are naturally high in iron, but mature seeds also have large amounts of phytic acid which inhibits absorption of minerals in the gut. We have shown that garden peas and petit pois, which are immature seeds, have accumulated lots of iron at this stage of development, but not yet much phytic acid. The higher iron-to-phytate ratio correlates with better iron bioavailability from garden peas compared to mature peas (Moore et al., 2018). Mature peas, also known as dry peas or marrowfat peas, are used in dishes such as ‘mushy peas’, peas pudding and Dutch-style pea soup.
The iron in peas is mostly stored in ferritin, a natural nanoparticle with an iron core of 2000-2500 iron atoms surrounded by a protein shell. Purified ferritin is highly bioavailable and we found that it can be taken up by different pathways in intestinal cells (Perfecto et al., J. Nutr, 2018).
We are also investigating the genetic basis of iron accumulation in peas, in lines from special collections (so called genetic variety panels) or lines with induced mutations. Recently, we have identified the underlying mutations of dgl and brz. These iron-accumulating mutants were identified more than 30 years ago and have contributed greatly to our understanding of iron homeostasis in plants before Arabidopsis took over as a model organism.
dgl is a small in-frame deletion in a BRUTUS homologue, an E3 ubiquitin ligase which suppresses activity of transcription factors for iron uptake; brz is a point mutation causing Leu466Phe in OPT3, an oligopeptide transporter required for xylem-to-phloem transport of iron (Harrington et al., BioRxiv 2023).