IND1 / NUBPL in complex I assembly and disease
Ind1 is a mitochondrial protein found in most eukaryotes, but it is absent from baker’s yeast (Saccharomyces sp.).
The ~30-kDa P loop ATPase was first characterized in the yeast Yarrowia lipolytica, where it is involved in the assembly of respiratory complex I, NADH:ubiquinone oxidoreductase (Bych et al., 2008b). The rather specific role in complex I assembly was later confirmed in human HeLa cells (Sheftel et al., 2009) and in the model plant Arabidopsis (Wydro et al., 2013a).
It has been suggested that Ind1 inserts FeS clusters into complex I, but this has not been demonstrated to date.
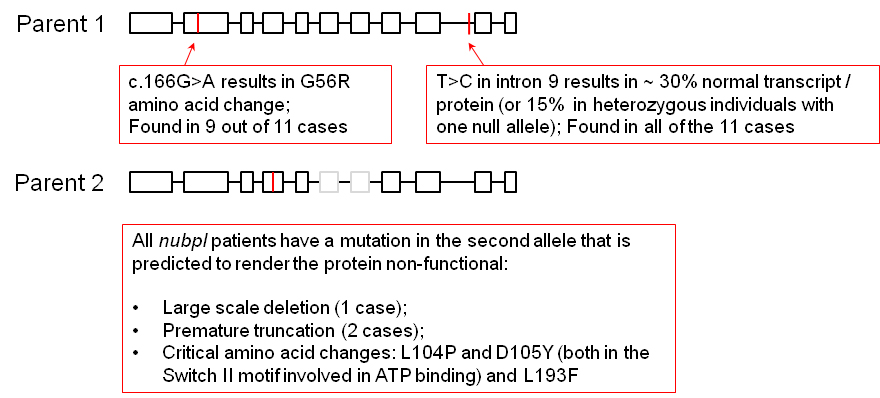
Mutations in the human homologue NUBPL (NUcleotide Binding Protein-Like) cause rare cases of encephalopathy (Calvo et al 2010 Nat. Genet.; Tucker et al 2012 Hum. Mutat.; Kevelam et al 2013 Neurology). All 15 cases identified so far are compound heterozygous for a specific splice-site mutation (c.815-27T>C) in combination with a null allele.
The splice-site mutation still allows a proportion of normal NUBPL protein to be formed. The Balk lab are using the yeast Yarrowia lipolytica as a model for testing pathogenicity of mutations (Wydro et al., 2013b; Maclean, PhD thesis).
Life without complex I in mistletoe, a hemi-parasitic plant
Respiratory complex I is essential for viability of the obligate aerobic yeast Yarrowia lipolytica. However, Baker’s yeast can live without, as it has lost complex I during evolution.
In the model plant Arabidopsis thaliana, knockout mutants of genes for key subunits of complex I, or for the INDH assembly factor, are much delayed in early development, but they are viable (Wydro et al., 2013). Still, it was thought that such mutants would not survive in the wild.
Surprisingly, the hemi-parasitic plant mistletoe completely lacks complex I, and it is the first multicellular organism in which this has been found (Maclean et al., 2018; Senkler et al., 2018). The loss of nad genes from the mitochondrial genome, which encode some of the complex I subunits, had previously been noticed (Skippington et al., 2015 PNAS 112, E3515; Petersen et al., Mol Biol Evol 31, 2586).
The respiratory chain of mistletoe mitochondria is extensively remodelled, with increased levels of alternative dehydrogenases and oxidases, and very low amounts of ATP synthase. Similar to Baker’s yeast, mistletoe obtains energy mostly from glycolysis and not from mitochondrial respiration (Maclean et al., 2018).

