We recall the landmark discovery of the first hybrid antibiotic
On Friday 14 September 1984, Professor David Hopwood announced to the world that his team at the John Innes Institute had produced the world’s first hybrid antibiotic by genetic engineering. David’s lab, in collaboration with a Japanese research group, had produced the antibiotic mederrhodin A. This breakthrough has been described by David as the closest he got to a ‘eureka moment’ in his career.
Throughout his career, his work had centred on the genetics and microbiology of a strain of soil-dwelling bacteria, Streptomyces coelicolor, which gets its name from the pigment it secretes. This sky-blue chemical compound is called actinorhodin, an antibiotic produced naturally to help S. coelicolor compete with other microbes in the soil.
For many years, the key to finding new antimicrobial drugs was to look among the natural products of the soil. By 1984, streptomycetes had yielded more than 70 commercially important antibiotics, and it was thought that most of what the soil had to offer had been discovered and put into use. The hunt for new drugs was becoming harder and harder.
David’s “hybrid antibiotic” breakthrough offered an alternative approach, opening up the possibility of making ‘unnatural natural products’ to extend the range of available compounds.
Genes for antibiotic production were first cloned from the ‘parent’ strain of Streptomyces coelicolor. The cloned genes were introduced into another streptomycete, one that usually makes a brown-coloured antibiotic called medermycin.
The genetically engineered Streptomyces then began to secrete a new, purple-coloured antibiotic (mederrhodin A) – made by both sets of genes working in ‘collaboration’. Spectroscopic analysis by Professor Satoshi Ōmura’s group at the Kitasato Institute, Tokyo, confirmed its hybrid chemical structure.
The appearance of the purple colour was David’s ‘eureka moment’; the experiment had shown how to create new compounds by transferring genes between organisms that make different compounds.
Scientists had already mastered getting one species of bacteria to manufacture a compound normally made by another species (by gene transfer); indeed, David’s lab had first demonstrated the cloning of the complete set of actinorhodin genes in this way. However, producing a totally novel compound by genetic engineering, was new and exciting. With this experiment, a new pharmaceutical field of ‘unnatural natural products’ was born.
In a second collaboration, an American group led by Professor Heinz Floss at Ohio State University, Columbus, had characterised a second hybrid antibiotic made by a strain arising when David’s group had transferred actinorhodin genes from S. coelicolor into a culture making another blue compound called dihydrogranaticin. However, this did not give rise to a change in colour, so it was not as dramatic as the mederrhodin case.
The formal announcement of the hybrid antibiotic discovery was made in Nature in April 1985 , recording the contributions of DA Hopwood, F Malpartida, HM Kieser, H Ikeda, J Duncan, I Fujii, BAM Rudd, HG Floss and S Ōmura. That landmark was the catalyst for the introduction of genetic approaches that transformed natural product chemistry.
What are streptomycetes?
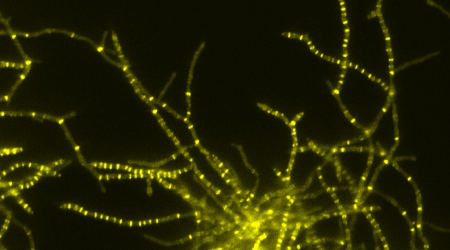
Streptomycetes are a group of soil-dwelling bacteria that have fascinated and confused biologists for many years after their discovery in the late-19th century because they appeared to be a mix between bacteria and fungi.
That taxonomic question was solved in the 1950s when it was confirmed that they are true bacteria and only resemble fungi superficially, growing in mycelial colonies with aerial hyphae and spores. To the naked eye they have a fuzzy appearance like moulds when grown on an agar plate.
Streptomycetes are actinomycetes, which secrete antibiotics to help them compete with other microorganisms living in the soil. In 1943, streptomycin was discovered and provided the first effective treatment for tuberculosis.