Sweet salvation – how a sugar cane pathogen is gearing up a new era of antibiotic discovery

A potent plant toxin with a unique way of killing harmful bacteria has emerged as one of the strongest new antibiotic candidates in decades.
The antibiotic, called albicidin, is produced by the bacterial plant pathogen Xanthomonas albilineans, which causes the devastating leaf scald disease in sugar cane. Albicidin is thought to be used by the pathogen to attack the plant, enabling its spread. It has been known for some time that albicidin is highly effective at killing bacteria including E. coli and S. aureus. These superbugs, notorious for their growing resistance to existing antibiotics, have prompted a vital need for effective new drugs.
Despite its antibiotic potential and low toxicity in pre-clinical experiments, pharmaceutical development of albicidin has been hampered because scientists did not know precisely how it interacted with its target, the bacterial enzyme DNA gyrase (gyrase) This enzyme binds to DNA and, through a series of elegant movements, twists it up (a process known as supercoiling) – a vital process for cells to function properly.
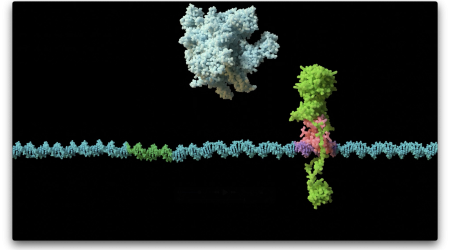
Now, Dr Dmitry Ghilarov’s research group at the John Innes Centre, alongside the laboratories of Prof. Roderich Süssmuth at Technische Universität Berlin, Germany and Prof. Jonathan Heddle at Jagiellonian University, Poland, have exploited advances in cryo-electron microscopy to obtain a first snapshot of albicidin bound to gyrase.
It showed that albicidin forms an L-shape, enabling it to interact with both the gyrase and the DNA in a unique way. In this state gyrase can no longer move to bring the DNA ends together. The effect of albicidin is akin to a spanner thrown between two gears.
The way albicidin interacts with gyrase is sufficiently different from existing antibiotics that the molecule and its derivatives are likely to be effective against many of the current antibiotic resistant bacteria.
“It seems by the nature of the interaction, albicidin targets a really essential part of the enzyme and it’s hard for bacteria to evolve resistance to that,” said Dr Ghilarov. “Now that we have a structural understanding, we can look to further exploit this binding pocket and make more modifications to albicidin to improve its efficacy and pharmacological properties.”
This work has already begun: the team used their observations to chemically synthesise variations of the antibiotic with improved properties. In tests, these variants were effective against some of the most dangerous hospital-acquired bacterial infections including Escherichia coli, Klebsiella pneumoniae, Pseudomonas aeruginosa and Salmonella typhimurium.
Dr Ghilarov said: “We believe this is one of the most exciting new antibiotic candidates in many years. It has extremely high effectiveness in small concentrations and is highly potent against pathogenic bacteria – even those resistant to the widely used antibiotics such as fluoroquinolones.”
“This molecule has been around for decades”, continued Dr Ghilarov, “Now advances in cryo-electron microscopy have made it possible to determine structures of even the most elaborate protein-DNA complexes. To be the first person to see the molecule bound to its target and how it works is a huge privilege, and the best reward one can have as a scientist. But this work is a big team effort, and we would not have done it without our European colleagues.”
The next step for this research is to engage with academic and industrial collaborators, and to seek funding to take the research forward to human clinical trials. This could lead to the development of an urgently needed new class of antibiotics in the face of a global threat of antimicrobial resistance, AMR.
Read the paper: “Molecular mechanism of topoisomerase poisoning by a potent peptide antibiotic” appears in Nature Catalysis. https://www.nature.com/articles/s41929-022-00904-1
Albicidin – how does it work?
Albicidin targets an enzyme found in both plants and bacteria called DNA gyrase (or simply “gyrase”). This enzyme binds to DNA and, through a series of elegant movements, twists it up (a process known as supercoiling) – a vital process for cells to function properly. However, gyrase has an Achilles heel; to do its job it must momentarily cut the DNA double helix. This is dangerous, as broken DNA is lethal to the cell. Normally, gyrase quickly joints the two pieces of DNA back together again as it works, but albicidin prevents it from happening, resulting in broken DNA and bacterial death.
About Anti-Microbial Resistance (AMR)
Multi-drug resistant pathogens such as Escherichia coli, Pseudomonas aeruginosa and Salmonella typhimurium present a dangerous healthcare burden, exacerbated by the COVID-19 pandemic.
Infections by resistant pathogens are a leading cause of death in hospital intensive care units, with some strains becoming pan resistant. Gram-negative drug-resistant pathogens were a cause of 50,000 deaths in 2019.
Despite urgently needed new medicines to combat this threat, drug discovery programmes have yielded no new classes of antibiotics for several decades.
Web links
Ghilarov lab
@dghilarov
Suessmuth lab
https://twitter.com/Suessmuth_Lab
https://www.biochemie.tu-berlin.de/
Heddle lab
http://www.heddlelab.org/
@Heddlelab
Image Credit – Alina Kurokhtina.
Caption – A sculpture that represents albicidin binding pocket, antibiotic pill and Petri dish to symbolise a threat caused by bacteria