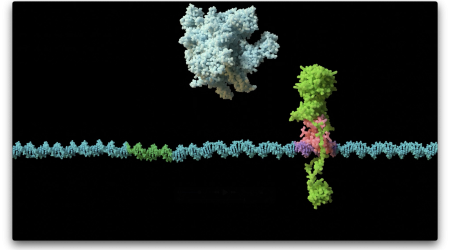
What is Antimicrobial Resistance (AMR)?
We spoke to Professor Tony Maxwell to find out, what is Antimicrobial Resistance, why is it a problem and how worried should we be?
“Antimicrobial Resistance (AMR) is the ability of a microbe (e.g. a bacterium) to survive antibiotic treatment. AMR also covers other microorganisms like fungi, which can be problematic in terms of human infections.
Simply put, we are running out of antibiotics
But what does that mean? We have plenty of antibiotics in our armoury after all, the problem is that a significant number of these are meeting a significant level of resistance.
It is a problem in the UK, particularly in hospitals, but it is a major problem across the globe, where some hospitals have a resistance rate of close to 100% to certain antibiotics, which means that a patient presenting at a hospital may not be treatable with the normal course of antibiotics.
In this country, in the 21st century, we have young, relatively healthy people who are going in to hospital, picking up a bug and then getting very sick and even dying. This shouldn’t happen, but it is because of a lack of effective antibiotics.
We are already experiencing bigger AMR outbreaks, which are costing thousands of lives each year. We don’t notice the effect in the UK, but there are diseases, like cholera, that are problematic elsewhere in the world.
If we had a serious AMR outbreak in the UK now we could be in deep trouble, because it takes so long to develop new treatments and we simply can’t wait that long.
The chances of an AMR epidemic are high: it is likely to happen; the timescale on when it happens we don’t know, it depends on so many factors.
It’s vital that people are more aware of this problem and we can all do our bit to limit the chances of this happening, such as not taking antibiotics unless they are needed and finishing each course of antibiotics you’re given.
Why is this occurring?
The simple answer is money. The current model for the production of antibiotics is that they are made, researched and developed by pharmaceutical companies, who then market them and, when they are prescribed, money is paid back to the pharmaceutical company.
Simply put, big-pharma companies are no longer able to make money from antibiotics, due to a number of factors that mitigate against this.
For example, as we all know, the more antibiotics we use, the less effective they are – that is not a good economic model. They are not a good prospect for companies, because they generally work. You get sick, you go to the doctor’s surgery and they give you a course of seven, or 10 days of antibiotics, which you take and recover, so you may never need to take them again; that’s no good financially.
A drug that makes you money, is something that people take long-term, ideally for the rest of their lives. This is not the drug companies’ fault. They are not charities and they are set up to make money for their employees and shareholders, which is a perfectly reasonable thing to do in a capitalist economy.
Further, if a drug company came up with an excellent antibiotic tomorrow, it would probably be put on the back shelf, because we need to ensure that we get the most mileage possible out of the antibiotics we already have, before we introduce new ones, because we know that resistance will inevitably follow; we cannot avoid resistance, we have to learn to live with it.
It is a constant fight with the bacteria, which ultimately the bacteria will win; they have been here a lot longer than us and will persist long after we are gone. Short-term, all we can do is keep the problem bacteria at bay – remember many bacteria are benign, or beneficial, such as those in our gut.
A major use of antibiotics is in agriculture. Fortunately, there has been a decrease in the use of antibiotics in agriculture, which tend to be used to prevent disease, rather than cure it. In terms of AMR this is a good thing and is being widely accepted across Western Europe, although the USA has not been quite as forthcoming.
I was shocked to read recently that antibiotics are being used in Florida orchards to combat citrus fruit diseases, which means those antibiotics may enter the food chain, either through the fruit, or through the soil. The worst thing you can do is expose bacteria to low doses of antibiotics as that encourages the development of resistant organisms so when you do need the antibiotics to treat disease, there is less chance of them working.
Where will the new antibiotics come from?
The good thing about antibiotics is that they are small(ish) chemical molecules that can generally be synthesised, produced and tolerated by humans.
There are a number of alternatives to antibiotics such as vaccines and phage therapy, which should also be investigated further. However, they all take time, up to a decade or two, to go from lab through clinical and non-clinical trials, before they can be used.
What we need is an alternative to pharmaceutical companies, and this is entirely feasible, in that all the expertise exists in other sectors. The entire process from discovery, all the way through to delivery could be covered, including all the research and development (R & D). But it is a long process and it is expensive.
It is estimated that it costs around $1 billion to bring a single antibiotic to the market, which sounds like a lot of money, but isn’t when you consider that there are a number of antibiotics which gross many billions per annum, so you can, in principle, make money from antibiotics. But of course, many fail, after a lot of money has already been invested.
It seems to me that one answer is government-funded antibiotic R & D. This may sound radical, given it would cost a lot of money, but the fact is that governments pay for antibiotics at some point. Currently we pay the pharmaceutical companies, who sell them back to us at the end of the process, but there is no reason they could not become the development company and pay for them at the beginning, doing the development ourselves.
If governments were prepared to pitch in significant sums of money, we could put together our own drug pipelines and link up the expertise that already exists in different places. This is currently being done in a European context, by the ‘Innovative Medicines Initiative’ (IMI) which is putting money into consortia to do this, but you can’t come to one place, such as the John Innes Centre, or the University of East Anglia and find that expertise all together.
Even on the Norwich Research Park, which has around 40 different groups working on anti-microbial resistance, we don’t have a joined-up way of bringing the appropriate expertise together. A pipeline needs to be built and then people recruited into it.
A lab like mine typifies the problem. We work on a tiny slice of a very big cake. That cake is antibiotic discovery, and within that we work on a group of enzymes found in bacteria, which are a very good target for antibiotics. It is pretty much luck that it happened.
We initially looked at them because they were interesting, in that they revealed some novel mechanistic principles, from a biological chemistry standpoint. It is a matter of good fortune that they turned out to be good targets for antibiotics.
We don’t always know how existing antibiotics work, just that they do, so identifying why and how they work is vital in allowing us to consider how others might work, or alternative ways we could attempt to make them work in order to bypass resistance.
For example, we are currently working on compounds with colleagues in Leeds University, that are effective in eliminating the target pathogen, but once the work reached trial stages they were found to be toxic to mice. This is the end of the road as far as pharmaceutical companies are concerned, but not for us, as we then try to look at how it works on a molecular level to see if we can do something to keep the effectiveness but remove the toxicity.
Many companies have developed antibiotics over the years and most of those end in failure, but that is no reason to throw all that work away because it may not be a bad antibiotic, it might just be one that won’t make the company any money. Given the seriousness of the current situation, we shouldn’t be worried about making money.
That’s why we had an Antibiotic Action intern who worked with other interns based with Professor Laura Piddock at the University of Birmingham, to provide information for an Antibiotic Database (AntibioticDB). To find out what compounds had been in the pipeline and, for one reason or another, not been developed, and then if there is any potential to take any forward now, scientific literature and abstracts from meetings were trawled through and phone calls to retired industry experts made.
With BSAC, Professor Piddock has made it free-at-the-point-of-use and it is searchable by many criteria including: compound name, drug company name, disease name, bacteria name etc, so you can explore what has been done and see if there are compounds which, using the expertise you have, you may be able to develop into drugs of the future.
Public-funded research and development is not a straightforward solution and it is not a cheap solution, but it probably is cheaper than paying pharmaceutical companies to do the same thing. My concern is that the time to do this is now, not in five or 10 years’ time; if we wait, it could be too late.”